|
9/13/2023 0 Comments Xe electron configurationCs⁺, as both ions have the same electron configuration as xenon (Xe). All of these: Since Sn⁴⁺ is not isoelectronic with Xe, this option is incorrect. When it loses 4 electrons to form Sn⁴⁺, it will have 46 electrons, which is not the same as Xe.ĭ. Sn⁴⁺: Tin (Sn) has an atomic number of 50, so it has 50 electrons in its neutral state. When it loses 1 electron to form Cs⁺, it will have 54 electrons, which is the same as Xe.Ĭ. Cs⁺: Cesium (Cs) has an atomic number of 55, so it has 55 electrons in its neutral state. Electron configuration can be done in two ways. We know that xenon's full electron configuration is 1s2\2s2\2p6\3s2\3p6\3d10\4s2\4p6\4d10\5s2\5p6 From this picture, we see that the next shell we choose to fill will be the 6s orbital. When it loses 2 electrons to form Ba²⁺, it will have 54 electrons, which is the same as Xe.ī. The electron configuration of europium is 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10 4s 2 4p 6 4d 10 4f 7 5s 2 5p 6 6s 2, if the electron arrangement is through orbitals. There are 54 electrons in an atom of xenon and the electronic configuration of an atom of xenon is 1 s 2 2 s 2 2 p 6 3 s 2 3 p 6 4 s 2 3 d 10 4 p 6 4 d 10 5. So, we start by writing the first part of tungsten's electron configuration which is Xe Next, we have 74-5420 more electrons to fill. Ba²⁺: Barium (Ba) has an atomic number of 56, so it has 56 electrons in its neutral state. The electron configuration for the first 10 elements. To find an ion that is isoelectronic with Xe, we need to find an ion that has the same number of electrons as Xe, which is 54 electrons. Electron Configurations are an organized means of documenting the placement of electrons based upon the energy levels and orbitals groupings of the periodic table. How do you write the electron configuration for Xenon The. Xenon (Xe) has an electron configuration of 1s² 2s² 2p⁶ 3s² 3p⁶ 4s² 3d¹⁰ 4p⁶ 5s² 4d¹⁰ 5p⁶. The electronic configuration of Xenon will be 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 5s2 5p6. The structure of Xenon hexafluoride is shown below.26. It donates its six electrons to six fluorine atoms to form six bonds and two electrons are left in the valence shell. This gives us: Xe 6s2 4f14 5d9 This notation is called the noble gas configuration, because the elements in the last column are the noble gases. 2 An element that has the same ground state valence-shell electron configuration as thallium is a. It has 8 valence electrons in its outermost electronic configuration so it can share its 8 electrons with other atoms.Īs in xenon hexafluoride xenon is bonded with six fluorine atoms. 1 Which of the following elements is found in the d-block of the periodic table Ir Tb Li Cl None of these Answer Exercise 7.3. The xenon atom has a total of 54 electrons so, we have to put 54 electrons in orbitals. For each atom the subshells are given first in concise form, then with all subshells written out, followed by the number of electrons per shell. 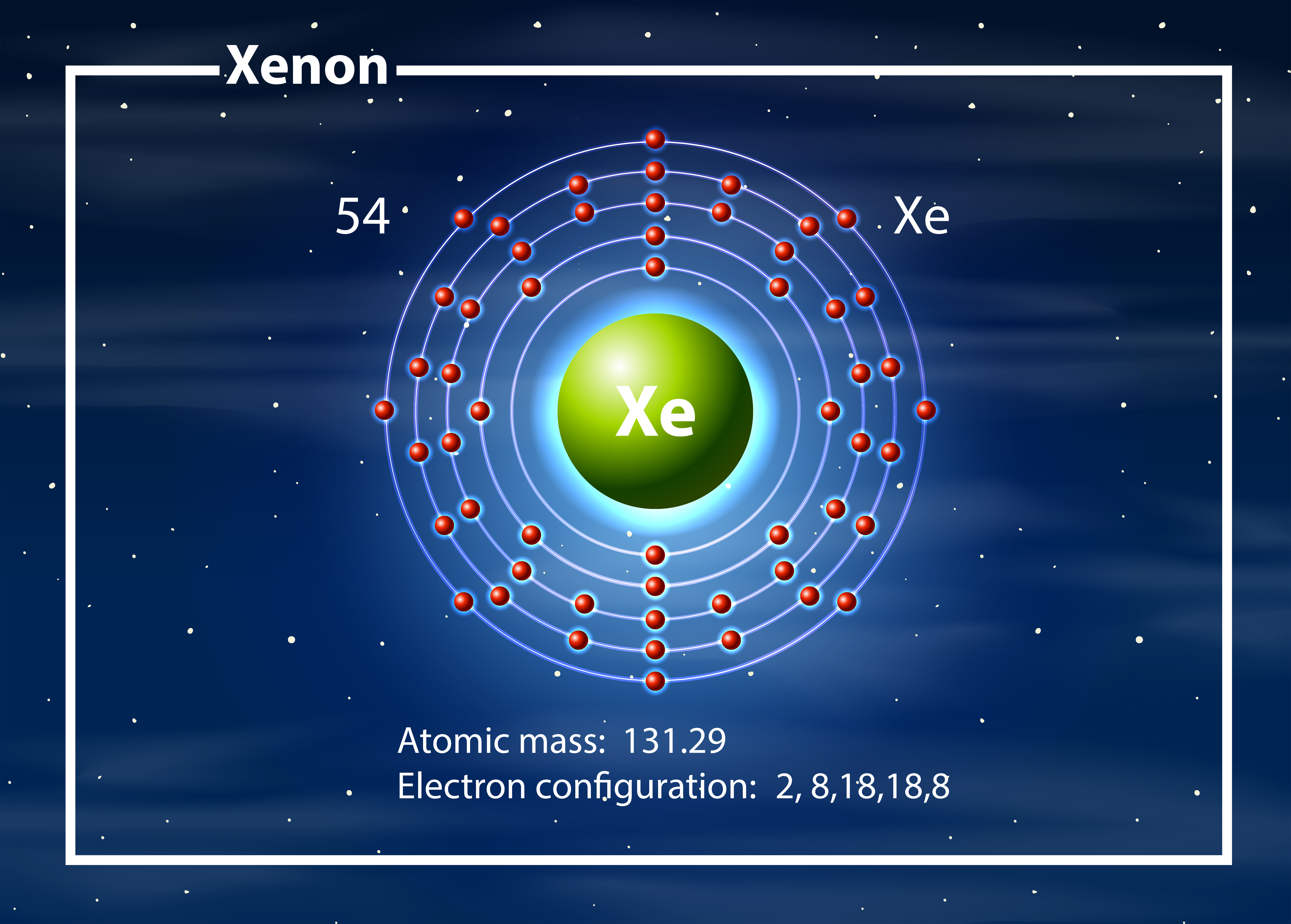
The notation X indicates that all subshells. Electron configurations of the elements (data page) This page shows the electron configurations of the neutral gaseous atoms in their ground states. 
The electronic configuration of xenon is $4$. Electron configuration is the arrangement of electrons on the orbitals. The ground electronic configurations of the elements H through U and their first cations are given below. Hint:The chemical element xenon is represented by Xe.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
